Special Interest Group Update
In each issue, one of NANN's special interest groups shares information in their area of focus.
Twin-to-Twin Transfusions
Tosha Harris, APRN DNP NNP-BC
Multiple-gestation pregnancies carry perinatal risks for both the gestational carrier and the fetuses. Complications such as twin-to-twin transfusion syndrome (TTTS), twin anemia polycythemia sequence (TAPS), and twin reversed arterial perfusion (TRAP) sequence are unique to multiple gestations, specifically monochorionic gestations.
Determining chorionicity and amnionicity is critical when managing multiple-gestation pregnancies. Monochronic twins are at higher risk for complications such as TTTS, TAPS, and TRAP sequence. Here are a few basic definitions to remember:
- Ovum: a mature female egg cell capable of being fertilized by a male sperm cell
- Zygote: a fertilized egg cell
- Zygosity: the type of conception; monozygotic twins develop from the division of one zygote. Dizygotic twins develop from the near synchronous fertilization of two ova by two sperm.
- Chorion: the outermost protective membrane that surrounds a fetus. The chorionic villi (vascular, finger-like projections) develop into the placenta (Davis, 2021)
- Chorionicity: the number of chorionic membranes surrounding the fetuses. Monochorionic twins share one chorion, and dichorionic twins have separate chorionic sacs.
- Amnion: the innermost protective membrane that surrounds a fetus. The amnion contains the amniotic fluid.
- Amnionicity: the number of amniotic sacs; monoamniotic twins share one amnionic sac, and diamniotic twins have separate amnionic sacs.
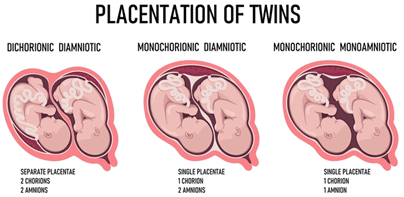
The chorionicity and amnionicity of multiple gestations produce three types of twins:
- Dichorionic–diamniotic twins have their own chorions and amniotic sacs; they typically do not share a placenta.
- Monochorionic–diamniotic twins share a chorion but have separate amniotic sacs; they share a placenta.
- Monochorionic–monoamniotic twins share one chorion and one amniotic sac; they share a placenta.
©Can Stock Photo Inc./Artinspiring
Monochorionic twin pregnancies have been associated with higher perinatal mortality and a higher incidence of preterm birth, low birth weight, and prolonged stay in the neonatal intensive care unit compared with dichorionic twin pregnancies (DePaepe, 2019). Additionally, monochorionic twin pregnancies are susceptible to TTTS, TAPS, TRAP sequence, and discordant growth restriction.
Etiology
TTTS results from chronic, unidirectional, and unbalanced blood transfusion from one twin (donor) to the other twin (recipient) through placental vascular connections. The primary etiology of TTTS is an increase in arteriovenous anastomoses in the cotyledon portion of the placenta (Borse & Shanks, 2022). If this flow imbalance is significant and not compensated by bidirectional arterio-arterial anastomoses (considered to be protective against TTTS), the donor becomes hypovolemic (oligiohydramiotic) and anemic, and the recipient becomes polycythemic and hypervolemic (polyhydramniotic) (Borse & Shanks, 2022; DePaepe, 2019).
TAPS form of chronic twin-to-twin transfusion is characterized by the significant difference in intertwin hemoglobin and reticulocyte levels in the absence of oligohydramnios and polyhydramnios. Small anastomoses allow for the slow transfusion of red blood cells from the donor twin to the recipient twin. This slow transfusion process provides time for hemodynamic compensation and thereby prevents the development of amniotic fluid imbalances. TAPS is characterized by a severe discordance in hemoglobin levels, whereas TTTS is characterized by a severe discordance in amniotic fluid levels (DePaepe, 2019).
TRAP sequence is a rare and severe form of twin-to-twin transfusion. While the exact pathogenesis of this condition remains debatable, the condition has two distinct components. First, there is a lack of cardiac development or function in the acardiac, malformed twin. Circulatory failure can be caused by hypoxia, the intrauterine demise of one twin, and fertilization of the polar body. Second, at least one arterio-arterial and usually one venovenous anastomosis must be present to support the development of the acardiac twin (DePaepe, 2019). The acardiac twin depends on the donor or pump twin for all its blood supply. The pump twin shunts part of its cardiac output toward the acardiac twin through retrograde flow through either the umbilical artery or arterio-arterial anastomoses. The reversed circulation of deoxygenated blood directly into the acardiac twin’s abdominal aorta preferentially perfuses the acardiac twin’s lower body and contributes to the anomalies characteristic of acardiac fetuses (DePaepe, 2019; Unal & Newman, 2021).
Diagnosis
Serial fetal ultrasound assessments make a diagnosis of twin-to-twin transfusion (Papanna & Bergh, 2022). TTTS is classically diagnosed by oligohydramnios in one amniotic sac and polyhydramnios in the other. The oligohydramnios deepest vertical pocket usually measures 8 cm (Unal & Newman, 2021).
In 1999, Quintero and colleagues developed a staging that categorizes the severity of TTTS cases:

Običan & Odibo, 2019; Papanna & Bergh, 2022; Unal & Newman, 2021
These criteria reflect the characteristics of the disorder in its truest form. However, staging can be challenging because the progression of cases is not always sequential. For example, cases can progress directly from stage 1 to stage 4, 5, and can exhibit stage 3 findings from the beginning.
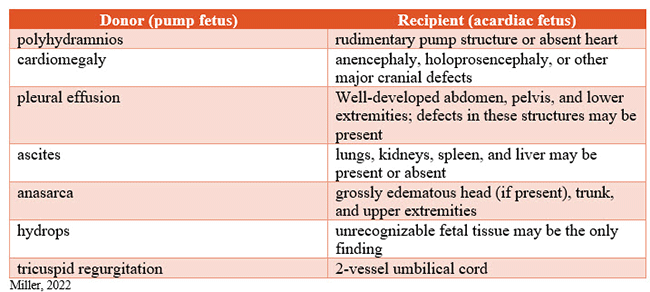
Ultrasound findings of retrograde blood flow through an arterio-arterial anastomosis are necessary to make a TRAP sequence diagnosis, and Doppler findings will show blood flowing toward, rather than away from, the acardiac twin. Other sonographic findings are listed in the table below.
Umbilical vein sampling is the confirmatory test used to diagnose TAPS. However, this testing poses a risk to the gestational carrier and the fetus. Using ultrasound to assess for anemia and polycythemia is the most common method to diagnose TAPS. Anemia and polycythemia are indicated when tests show a middle cerebral artery-peak systolic velocity (MCA-PSV) of >1.5 multiples of median (MoM) in one twin (suggestive of anemia) and 1.7 (reticulocyte count of the donor twin divided by the reticulocyte count of the recipient twin)” (Papanna & Bergh, 2022).
Management
The North American Fetal Therapy Network (NAFTNet), the Society for Maternal-Fetal Medicine (SMFM), and the American College of Obstetricians and Gynecologists (ACOG) have recommended evaluating ultrasounds every 2 weeks for monochorionic gestations beginning at 16 weeks’ gestation and continuing until delivery (Borse & Shanks, 2022; Unal & Newman, 2021).
SMFM recommends delivery timing around 34 to 37 weeks if possible. To promote fetal lung maturation, SMFM also recommends considering steroids between gestational ages of 24 to 34 weeks, primarily due to the higher risk of preterm birth in this population.
Several modalities have been used to treat twin-to-twin transfusion, including expectant management, amnioreduction, and laser photocoagulation. Selective reduction and voluntary pregnancy termination could once be offered as treatment options; however, the recent overturning of Roe v. Wade by the United States Supreme Court has made these options less likely to be offered. The most commonly used therapies target the unequal fluid levels between the twins’ amniotic sacs or interrupt the blood vessel communications, and recommendations are often based on staging and gestation.
Expectant management is recommended for stage 1 of TTTS as the prognosis for this stage is promising without further treatment. Fifteen percent to twenty-five percent of stage I cases will likely progress to a more advanced stage when managed expectantly (Borse & Shanks, 2022; Unal & Newman, 2021). Therefore, close monitoring with every 2-week scheduled ultrasounds is imperative.
Laser photocoagulation is the preferred treatment of choice in gestations earlier than 26 weeks and for TTTS stage 2 or greater. In this procedure, the goal is to ablate all anastomosing vessels connecting the fetuses. Amnioreduction is considered in cases where laser photocoagulation cannot be performed due to advanced gestational age or technical difficulties. An amnioreduction is typically performed to correct the polyhydramnios to 14 weeks’ gestation. Amnioreductions can be performed once or serially (Borse & Shanks, 2022; Unal & Newman, 2021).
“The management of TAPS is typically expectant with intervention offered for persistent and worsening stage 2 or higher TAPS. Choice of intervention is based on gestational age, disease stage, and physician expertise. Persistent advanced stage TAPS beyond 32 weeks is managed with delivery” (Papanna & Bergh, 2022).
TRAP sequence treatment is aimed solely at preserving the life and optimizing the outcomes of the pump twin. Umbilical cord occlusion therapy is the treatment of choice for TRAP sequence and can be accomplished by radiofrequency ablation (RFA), bipolar cord coagulation, and intrafetal (or interstitial) laser coagulation. The most commonly used technique is RFA; however, the technique of choice should be based on operator experience, center resources, clinical presentation, gestational age, and preference. Cord occlusion therapy interrupts the blood vessel communications between the acardiac twin and the pump twin; this allows for hemodynamic stabilization of the pump twin and the ultimate demise of the acardiac twin. Due to the need for highly specialized training and equipment, only a small number of institutions offer this procedure (Miller, 2022).
Multiple-gestation pregnancies account for 3% of all births. According to DePaepe (2019), the increase in occurrence can be attributed to older maternal age distribution and the increased use of assisted reproductive technologies (ART) and non-ART therapies. Twin gestations carry a higher risk for complications than singleton gestations, and monochorionic twins comprise 20%–25% of all twin pregnancies (DePaepe, 2019). Monochorionic twins are at higher risk for complications such as TTTS, TAPS, and TRAP sequence. Given the high prematurity rates of these infants, neonatal clinicians are encouraged to remain current on the most recent evidence-based literature regarding this phenomenon.
References
Borse, V., & Shanks, A. L. (2022). Twin-to-twin transfusion syndrome. StatPearls. http://www.ncbi.nlm.nih.gov/books/NBK563133/
Davis, C. P. (Ed.). (2021, March 29). Medical definition of chorion. MedicineNet. Retrieved July 22, 2022, from https://www.medicinenet.com/chorion/definition.htm
DePaepe, M. E. (2019). Multiple gestation: The biology of twinning. In R. Resnik, C. J. Lockwood, T. R. Moore, M. F. Greene, J. A. Copel, R. M. Silver (Eds.), Creasy and Resnik’s maternal-fetal medicine: Principles and practice, (8th ed., pp. 68–80). Elsevier.
Miller, R. (2022). Diagnosis and management of twin reversed arterial perfusion (TRAP) sequence. In D. Levin, L. L. Simpson (Eds.), UptoDate. Retrieved November 16, 2022, from https://www.uptodate.com/contents/diagnosis-and-management-of-twin-reversed-arterial-perfusion-trap-sequence?search=acardiac%20twin%20diagnosis&source=search_result&selectedTitle=1~8&usage_type=default&display_rank=1#H3361529130
Običan, S. G., & Odibo, A. O. (2019). Invasive fetal therapy. In R. Resnik, C. J. Lockwood, T. R. Moore, M. F. Greene, J. A. Copel, R. M. Silver (Eds.), Creasy and Resnik’s maternal-fetal medicine: Principles and practice, (8th ed., pp. 594–631). Elsevier.
Papanna, R., & Bergh, E. (2022). Twin anemia-polycythemia sequence (TAPS). In D. Levin, L. L. Simpson (Eds.), UptoDate. Retrieved November 16, 2022, from https://www.uptodate.com/contents/twin-anemia-polycythemia-sequence-taps?search=TAPS%20treatment&source=search_result&selectedTitle=1~38&usage_type=default&display_rank=1#H710537235
Unal, E. R., & Newman, R. B. (2021). Multiple gestations. In M. B. Landon, H. L. Galan, E. R. M. Jauniaux, D. A. Driscoll, V. Berghella, W. A. Grobman, S. J. Kilpatrick, A. G. Cahill (Eds.), Gabbe’s obstetrics: Normal and problem pregnancies, (8th ed., pp. 751–783). Elsevier.


