Special Interest Group Update
In each issue, one of NANN’s special interest groups shares information in their area of focus.
A Review of Common Neonatal Arrhythmias
Julie Williams, DNP CRNP NNP-BC
Neonatal nurses are responsible for identifying abnormal cardiac rates and rhythms and conveying their concerns to other providers. For the untrained eye, it can be challenging to identify which arrhythmias are benign and which are pathologic. The notion of identifying arrhythmias can be overwhelming for many nurses. This article will identify various neonatal arrhythmias and discuss the management of some common neonatal arrhythmias.
The first step to identifying abnormal heart rates and rhythms is to understand what is normal. The normal newborn heart rate ranges from 90 to180 bpm and should have consistent variability. A heart rate of 90 bpm is generally observed in the term neonate and may be abnormal in a preterm infant. A heart rate faster than 180 bpm can be normal for an infant depending on the activity level (i.e., fussy or crying). Determining whether a particular heart rate is normal for an infant requires an evaluation of the infant's clinical picture (including medications, confounding disease processes, among others) and baseline heart rate trends.
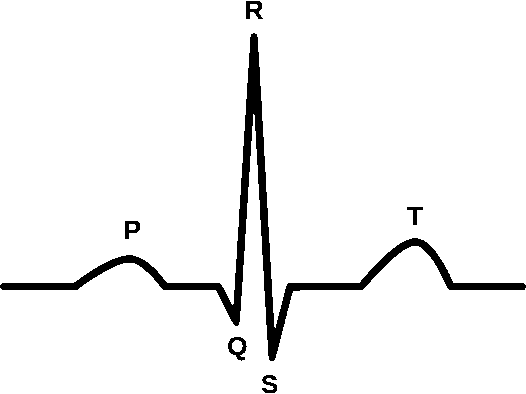
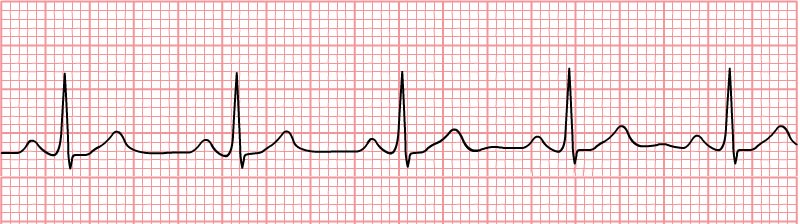
Sinus Rhythm
A normal sinus rhythm indicates atrial activation originating from the sinus node. Electrocardiographic tracings reflect a normal P wave before each QRS complex, a normal P wave axis characterized by upright P waves in leads I and II and inverted in augmented vector right (aVR). The PR interval should remain constant, and the QRS complex should be less than 100 ms wide.
Neonatal Arrhythmias
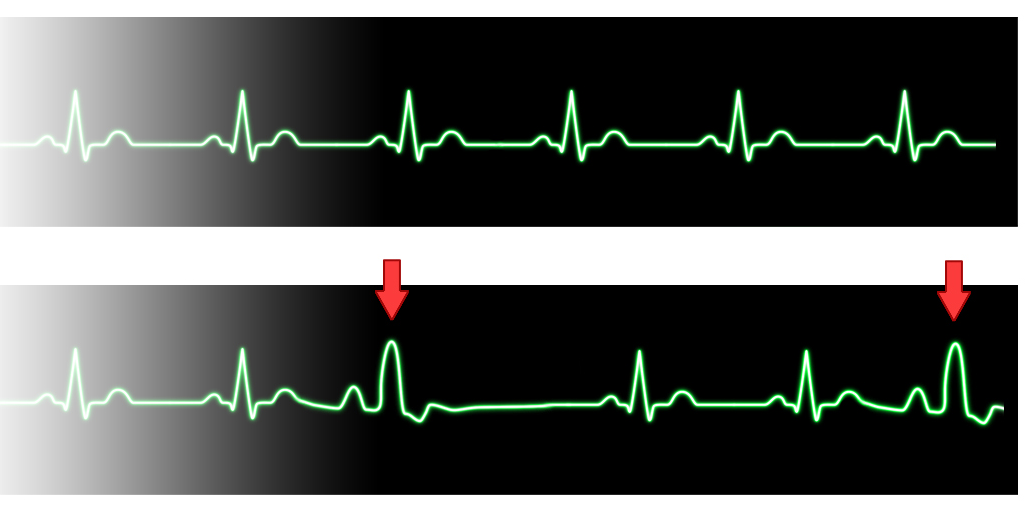
Premature Complexes
Premature complexes can arise from the atrium, ventricle, or atrioventricular (AV) junction. An occasional premature atrial contraction (PAC) or premature ventricular contraction (PVC) is common in the newborn. However, more frequent premature contractions require an evaluation and can be related to electrolyte imbalance, irritation from an intracardiac venous catheter, or structural heart disease (Baskar & Czosek, 2020). PACs are characterized by a premature P wave superimposed on the preceding T wave. The QRS morphology is similar to that seen in sinus rhythm (Ban, 2017). Occasionally, a wide QRS complex will be noted with a PAC; these complexes are called conducted PAC, indicating transmission of an electrical impulse to the ventricles. PVCs are not as common as PACs. PVCs are characterized by a lack of a P wave and a premature QRS complex. The morphology of the QRS complex is different from sinus rhythm (Ban, 2017). While an etiology for the PAC or PVC is generally unknown, a thorough evaluation of the electrolytes and evaluation for correct placement of any central lines and echocardiogram should be undertaken.
Premature Ventricular Contraction
Bradycardia
Bradycardia is another common arrhythmia. The more frequent classifications include normal sinus bradycardia and heart block. Sinus bradycardia can occur from a combination of ectopic foci in the atrium, ventricle, or AV junction (Baskar & Czosek, 2020). These ectopic foci can be evident in profound sinus bradycardia. The etiology of sinus bradycardia can include cardiorespiratory failure and neurological abnormalities.
Heart block signifies a disturbance of AV conduction. The etiology can be congenital, as seen with maternal systemic lupus erythematosus (SLE), or acquired, as seen with infection and post-surgery. Treatment will depend on the degree of heart block; options include placement of a pacemaker and medication in the case of an infection.
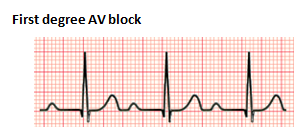
The severity and location of the AV conduction disturbance determine the degree of heart block. In a first-degree heart block, there is a prolonged PR interval without any other abnormalities. A PR interval of longer than 160 ms is necessary to confirm the diagnosis (Cannon & Snyder, 2020). First degree AV block is not a true block but a prolongation of AV conduction; it is usually benign and does not cause bradycardia. Etiologies include cardiomyopathy, congenital heart disease (i.e., Ebstein anomaly), and medications like digoxin (Cannon & Snyder, 2020). No treatment is indicated for first degree AV block. However, follow up may be advised in conditions of significant PR prolongation.
Second-degree heart block is characterized by intermittent AV conduction. In this disorder, some of the sinus beats are transmitted to the ventricles, but not all. There are two forms, Mobitz Type I and Mobitz Type II. Mobitz Type I, also referred to as Wenckebach phenomenon, is characterized by a progressive lengthening of the PR interval until one QRS is "dropped." The subsequent PR interval is normal. Mobitz Type II is associated with a stable PR interval with intermittent loss of conduction. This disorder is characterized by conduction disease within the His-Purkinje system and can precede a complete heart block (CHB). Mobitz Type I is usually benign and caused by medications and conditions associated with increased vagal tone. Mobitz Type II is rarely seen in the neonatal population. Patients with Mobitz Type II may require a pacemaker.
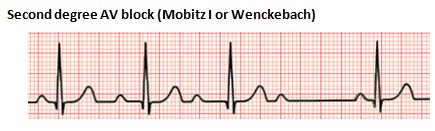
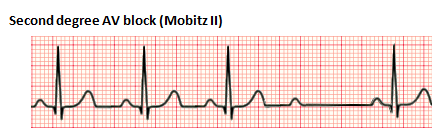
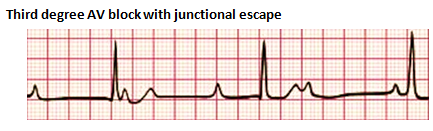
Third-degree heart block, also referred to as complete heart block, is characterized by the absence of any atrial electrical conduction to the ventricles due to an abnormality of the AV node, a bundle of His, or both bundle branches (Baskar & Czosek, 2020; Cannon & Snyder, 2020). There is typically ventricular activation related to the junctional or ventricular escape rhythm. However, the rhythm is generally slower than the sinus rhythm and does not have AV synchrony. CHB can be acquired or congenital. The acquired form can be autoimmune, idiopathic, a complication of cardiac surgery, or from certain congenital heart defects. Treatment may include the placement of a permanent pacemaker.
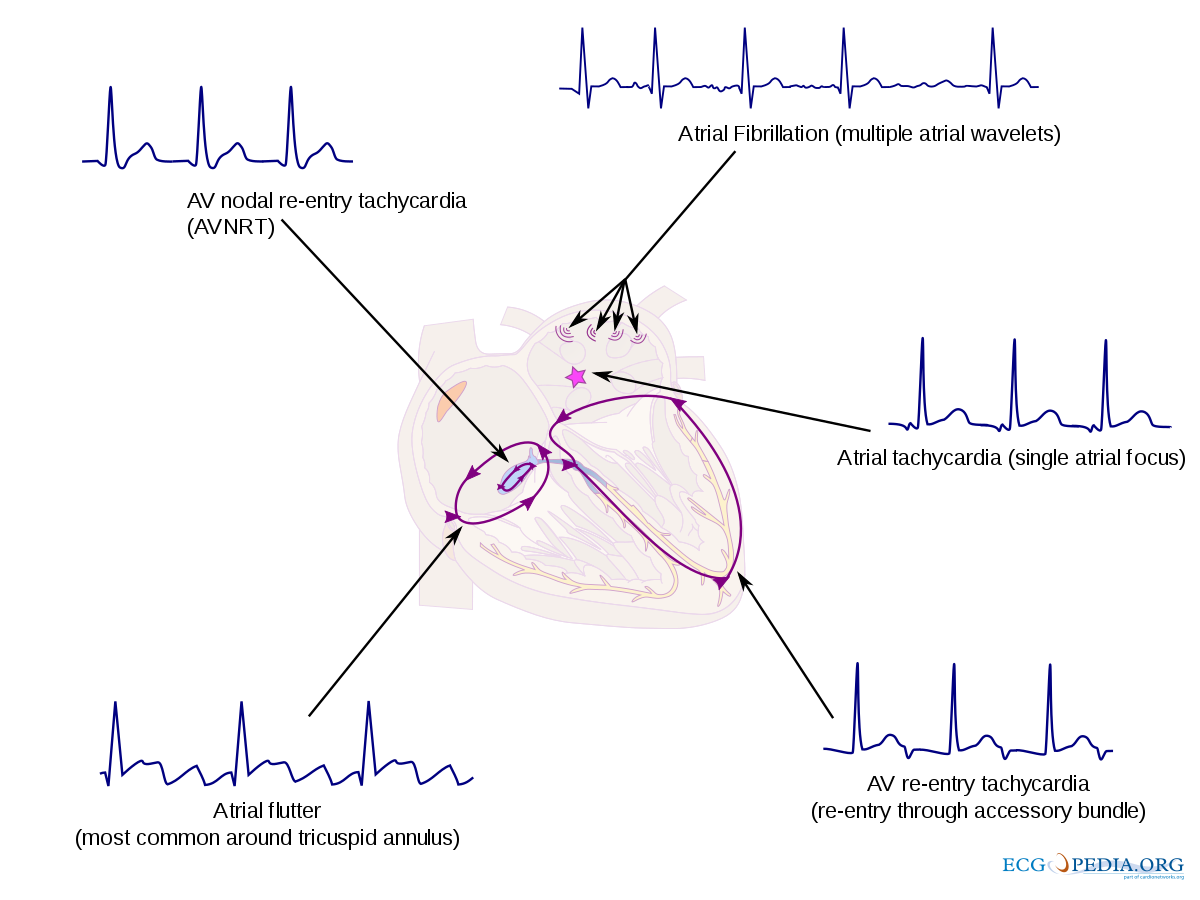
Tachycardia
Tachycardia includes supraventricular tachycardia (SVT) and ventricular tachycardia. Supraventricular tachycardia (SVT) is the most common tachyarrhythmia in the neonatal period. SVT is described as any tachycardia arising from or above the bundle of His (Baskar & Czosek, 2020). Many tachyarrhythmias fall under the category of SVT, including AV nodal reentrant tachycardia, atrial flutter, atrial fibrillation, atrial tachycardia, and AV reentry tachycardia.
In ventricular tachycardia, an abnormal rhythm originates in the ventricular myocardium or from the distal bundle of the His-Purkinje system below the bundle of His (Baskar & Czosek, 2020). The most common type of VT is an accelerated idioventricular rhythm (AIVR). Infants with AIVR are usually asymptomatic unless there are other cardiac anomalies. Treatment for tachycardia can vary from medications including adenosine or amiodarone to cardioversion. A thorough evaluation of the cardiac rhythm by a cardiologist is necessary to correctly identify the arrhythmia.
Neonatal arrhythmias can be a very frightening event for the neonatal nurse. Establishing a baseline understanding of which arrhythmias are benign versus pathologic is essential. A systematic approach to the evaluation of each arrhythmia can help ease the fear experienced.
References:
- Ban, J. (2017). Neonatal arrhythmias: Diagnosis, treatment, and clinical outcome. Korean Journal of Pediatrics, 60(11), 344-352. doi:10.3345/kjp.2017.60.11.344
- Baskar, S., & Czosek, R. J. (2020). Evaluation and management of common neonatal arrhythmias. Neoreviews (Elk Grove Village, Ill.), 21(9), e605-e615. doi:10.1542/neo.21-9-e605
- Cannon, B., & Snyder, C. (2020). Disorders of Cardiac Rhythm and Conduction in Newborns. In R. Martin, A. Fanaroff, & M. Walsh, Fanaroff and Martin's Neonatal-Perinatal Medicine (pp. 1375-1392). Philadelphia PA: Elsevier.
Images:
- https://upload.wikimedia.org/wikipedia/commons/8/89/Heart_block.png
- https://upload.wikimedia.org/wikipedia/commons/6/6f/AV_block_sequence_%28CardioNetworks_ECGpedia%29.png
- https://www.google.com/imgres?imgurl=https%3A%2F%2Fupload.wikimedia.org%2Fwikipedia%2Fcommons%2F1%2F13%2FPremature_Ventricular_Contractions.png&imgrefurl=https%3A%2F%2Fcommons.wikimedia.org%2Fwiki%2FFile%3APremature_Ventricular_Contractions.png&tbnid=N3_5E-QwZoFN3M&vet=12ahUKEwjMnO2npdvsAhWGdt8KHaYgC2kQMygCegQIARBF..i&docid=Fa9kos5Yh8Y5kM&w=1600&h=1000&q=premature%20ventricular%20complex&hl=en&client=firefox-b-1-d&ved=2ahUKEwjMnO2npdvsAhWGdt8KHaYgC2kQMygCegQIARBF
- https://upload.wikimedia.org/wikipedia/commons/thumb/e/e1/SVT_overview_%28CardioNetworks_ECGpedia%29.svg/1200px-SVT_overview_%28CardioNetworks_ECGpedia%29.svg.png
Please note: The information presented and opinions expressed herein are those of the authors and do not necessarily represent the views of the National Association of Neonatal Nurses. Any clinical or technical recommendations made by the authors must be weighed against the health care provider's own judgment and the accepted guidelines published on the subject.
Check out last issue's SIG Update on Neonatal Transfusion Therapy


